
Dr. David Blok
Assistant Professor, Erasmus School of Social and Behavioural Sciences, Erasmus University Rotterdam
Dr. Blok has contributed to leprosy research by developing and evaluating intervention strategies using infectious disease transmission models, combined with epidemiological and economic approaches, to better understand disease dynamics and inform decision-making for control strategies.
Editor’s note: As a modeler, Dr. Blok uses computer simulations to explore “what‑if”questions for leprosy programs. By combining real‑world data with mathematical models of how leprosy spreads in families and communities, he can estimate how many infections are still hidden, project how many new cases might appear in the future, and test how much different strategies – such as SDR-PEP, earlier diagnosis, or new diagnostic tools – could speed up progress towards interrupting transmission.
Evidence indicates that single-dose rifampicin (SDR) reduces the risk of developing leprosy among contacts by approximately 60%. SDR starts working immediately by killing the bacteria that cause leprosy in people who have been exposed. This helps prevent early infection from developing into disease. However, this benefit is not immediately reflected in the number of new leprosy cases reported in the community because of the disease’s long incubation period. It takes time before the protective effect becomes visible in overall case numbers.
SDR as post-exposure prophylaxis (SDR-PEP) is administered alongside intensified contact tracing and active screening. As a result, the number of reported leprosy cases may initially increase. This rise is typically attributable to the detection of a backlog of undiagnosed cases in an area. After this transitional phase, a more rapid decline in the number of new leprosy cases is generally observed compared with routine program scenarios without preventive intervention.
The impact of SDR-PEP increases progressively over time. As more contacts are screened and administered SDR-PEP, the cumulative preventive effect strengthens, leading to a faster decline in new leprosy cases. With sustained high coverage, this approach accelerates the reduction in incidence and supports earlier interruption of transmission.
Global modeling suggests that consistent, high-coverage implementation of combined contact screening and SDR-PEP worldwide could reduce observed cases by up to 90% within approximately 22 years. Achieving such impact would require screening and providing SDR to an estimated 40 million contacts of index patients worldwide over the next two decades.
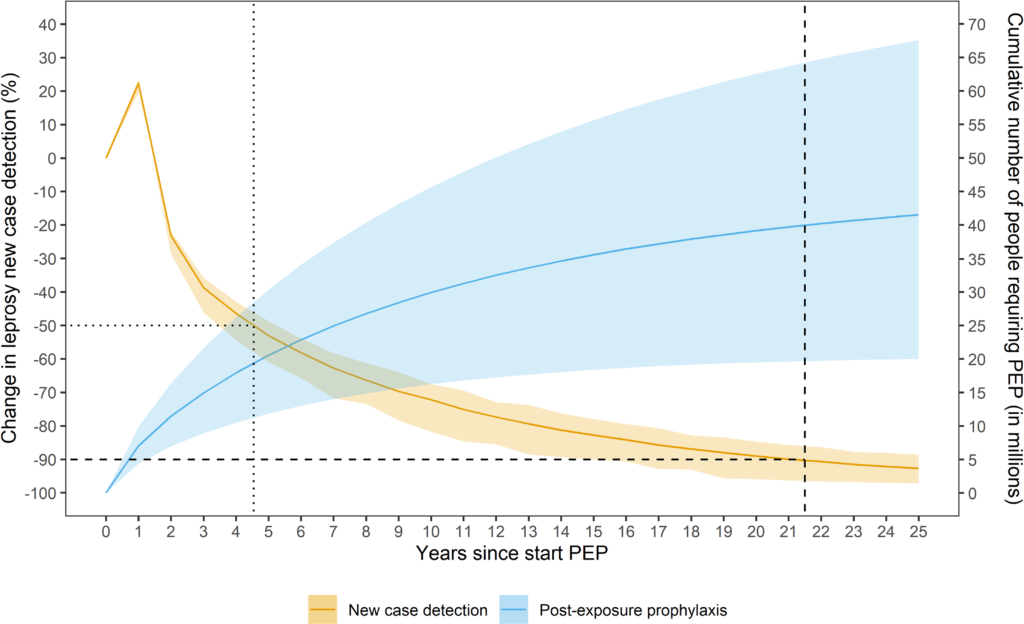
Projected change in new case detection and cumulative number of people requiring post‑exposure prophylaxis over 25 years. Reproduced from Taal AT, Blok DJ, van Brakel WH, de Vlas SJ, Richardus JH. Number of people requiring post‑exposure prophylaxis to end leprosy: A modeling study. PLoS Negl Trop Dis. 2021;15(2):e0009146. doi:10.1371/journal.pntd.0009146.
In reality, the impact of combined contact screening and SDR-PEP varies substantially across countries and settings. A key determinant is the number of contacts screened per index case (the first diagnosed person in the setting). Higher contact coverage leads to greater preventive impact. For example, during the multi-country leprosy post- exposure prophylaxis (LPEP) project, it was shown that districts with a high number of contacts included (>20 contacts per index patient) would yield the highest reduction in the number of new leprosy cases. This means that both household contacts and neighbors should be included.
Also, the level of endemicity influences outcomes. In highly endemic countries such as India, a larger number of contacts are identified, screened, and deemed eligible for SDR, resulting in a greater absolute reduction in cases compared with lower-endemic settings.
Furthermore, the impact is larger if the routine leprosy program is strong, which implies that there are fewer undiagnosed clinical leprosy patients in the community. Undiagnosed patients continue to contribute to transmission, making preventive interventions less efficient. At the same time, the introduction of SDR-PEP can strengthen program performance. By promoting active contact tracing, improving surveillance, and increasing community engagement, SDR-PEP can help reinvigorate routine services and contribute to greater program efficiency and impact over the longer term.









